Navigating the cosmetic product registration process in Indonesia can be complex. From understanding the required documents to anticipating potential delays, businesses often have many questions. At INSIGHTOF Consulting Indonesia, we specialize in helping cosmetic brands successfully enter the Indonesian market. Below, we address some of the most common inquiries based on our clients’ experiences.
Our Services
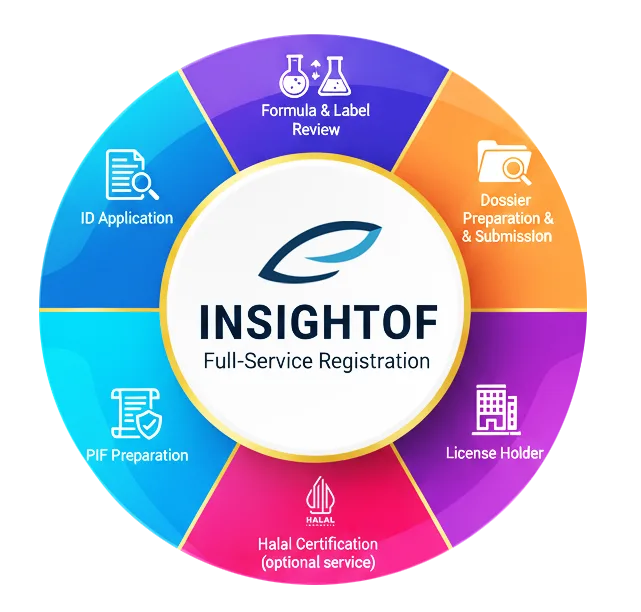
When you work with INSIGHTOF, our registration service is designed to cover the entire process from start to finish, including:
- ID application
- Formula and label review
- Dossier preparation and submission
In addition, we take on responsibilities such as acting as the license holder and preparing the Product Information File (PIF).
We also provide Halal certification services upon request, ensuring your products comply with Indonesia’s Halal Product Assurance regulations. Manufacture inspections, if related to Halal certification, are handled as part of this service.
While some companies only require formula or label reviews at the research stage, we do not provide these as standalone services. However, our team is happy to share guidelines and advice to support you in the early stages of product development.
Registration Timeline
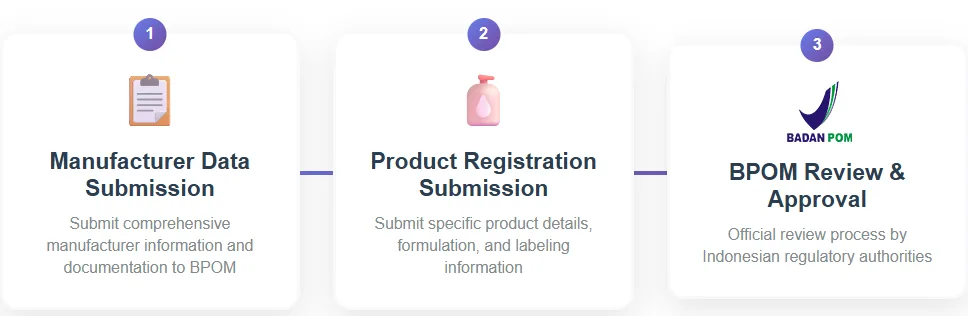
The standard timeframe for cosmetic registration in Indonesia is 3–6 months. This reflects two main stages of the process:
- Manufacturer data submission
- Product registration submission
Both stages are subject to BPOM’s review process and depend heavily on the completeness of the submitted documents. With well-prepared and complete documentation, registration can sometimes be finalized in as little as 1.5 months.
Key Considerations
Supplementary Documents
Occasionally, additional documents may be requested during the process. These requirements vary depending on your product and ensure compliance with BPOM’s regulations. At INSIGHTOF, we always provide clear explanations for any supplementary documents requested.
Uncommon Ingredients
If your product contains ingredients not listed in Indonesia’s database, these must first be registered before the cosmetic product itself can be approved. This step may extend the timeline but generally does not require additional testing.
Mandatory Testing Reports
For cosmetic notifications in Indonesia, mandatory testing reports are usually not required. However, specific formulas may have unique requirements, and we confirm these after reviewing your documentation.
INSIGHTOF Services
At INSIGHTOF Consulting Indonesia, our goal is to simplify the regulatory process so you can focus on growing your brand. If you’re planning to register cosmetic products in Indonesia, our team is here to guide you every step of the way.

Contact INSIGHTOF today for a free consultation. Our expert team will help ensure your products meet halal certification requirements and achieve a smooth, compliant entry into the Indonesian market.
Do you need assistance registering your product in Indonesia?
Contact us today to start your registration process.



.svg/240px-YouTube_social_red_squircle_(2017).svg.png)






